Welcome back, dear student! In Unit 1 we studied acid-base equilibria. Now in Unit 2, we will explore a very exciting topic — Electrochemistry. This is the branch of chemistry that deals with the relationship between electrical energy and chemical changes. Think about batteries that power your phone, or the process of electroplating jewelry — all of these depend on electrochemistry! Let’s learn it step by step.
2.1 Oxidation-Reduction Reactions
Before we talk about electrochemical cells, we need to understand the type of reactions that happen inside them. These are called oxidation-reduction (redox) reactions.
2.1.1 Oxidation
What comes to your mind when you hear “oxidation”? Many students think of iron rusting or a piece of paper burning. You are on the right track! But in chemistry, oxidation has a very precise meaning.
Remember: “LEO” — Loss of Electrons is Oxidation.
Let’s look at a simple example. When sodium metal reacts with chlorine gas:
2Na(s) + Cl2(g) → 2NaCl(s)
Let’s examine what happens to sodium. A neutral Na atom has 1 valence electron. In NaCl, sodium becomes Na+ — it has lost its electron!
Na → Na+ + e−
This loss of electrons is oxidation. The oxidation number of Na increases from 0 to +1.
2.1.2 Reduction
Now, what happens to chlorine in the same reaction?
Remember: “GER” — Gain of Electrons is Reduction.
Together: “LEO the lion says GER” — Loss of Electrons is Oxidation, Gain of Electrons is Reduction.
Chlorine gains the electron that sodium lost:
Cl2 + 2e− → 2Cl−
The oxidation number of Cl decreases from 0 to −1. This is reduction.
Important: Oxidation and reduction always happen TOGETHER. You cannot have one without the other. If something loses electrons, something else must gain them!
- Oxidation = loss of electrons = increase in oxidation number.
- Reduction = gain of electrons = decrease in oxidation number.
- The substance that is oxidized is called the reducing agent (it causes reduction of another substance).
- The substance that is reduced is called the oxidizing agent (it causes oxidation of another substance).
- In our example: Na is oxidized (reducing agent), Cl2 is reduced (oxidizing agent).
Practice Question 1: In the reaction Zn + CuSO4 → ZnSO4 + Cu, identify what is oxidized, what is reduced, the oxidizing agent, and the reducing agent.
Answer:
Zn → Zn2+ + 2e− (oxidation number increases from 0 to +2) → Zn is oxidized (reducing agent).
Cu2+ + 2e− → Cu (oxidation number decreases from +2 to 0) → Cu2+ is reduced (oxidizing agent).
Practice Question 2: Determine the oxidation number of Mn in KMnO4.
Answer: KMnO4 is neutral, so the sum of oxidation numbers = 0.
K = +1, O = −2 (each), let Mn = x
(+1) + x + 4(−2) = 0
1 + x − 8 = 0
x = +7
The oxidation number of Mn in KMnO4 is +7.
2.1.3 Balancing Oxidation-Reduction (Redox) Reactions
Balancing redox reactions is a very important skill for your exam. There are two main methods: the oxidation number method and the half-reaction (ion-electron) method. Let’s learn both carefully.
Method 1: Oxidation Number Method
Follow these steps:
- Assign oxidation numbers to all elements.
- Identify which elements are oxidized and which are reduced.
- Calculate the increase and decrease in oxidation numbers.
- Use coefficients to make the increase equal to the decrease.
- Balance the remaining atoms by inspection.
- Balance O by adding H2O and balance H by adding H+ (for acidic) or OH− (for basic).
Question: Balance the following redox reaction in acidic medium: MnO4− + Fe2+ → Mn2+ + Fe3+
Solution (step-by-step):
Step 1: Assign oxidation numbers:
Mn in MnO4−: x + 4(−2) = −1 → x = +7
Mn in Mn2+: +2
Fe in Fe2+: +2; Fe in Fe3+: +3
Step 2: Identify changes:
Mn: +7 → +2 (decrease of 5) → Reduction
Fe: +2 → +3 (increase of 1) → Oxidation
Step 3: Make increase = decrease. LCM of 5 and 1 is 5.
So: 1 MnO4− and 5 Fe2+
Step 4: Write the unbalanced equation:
MnO4− + 5Fe2+ → Mn2+ + 5Fe3+
Step 5: Balance O by adding H2O (left side has 4 O):
MnO4− + 5Fe2+ → Mn2+ + 5Fe3+ + 4H2O
Step 6: Balance H by adding H+ (right side has 8 H):
MnO4− + 5Fe2+ + 8H+ → Mn2+ + 5Fe3+ + 4H2O
Step 7: Check charge balance: Left = −1 + 10 + 8 = +17; Right = +2 + 15 + 0 = +17 ✓
- First find the total increase and total decrease in oxidation numbers.
- Use LCM to make them equal — this gives you the coefficients.
- Balance O with H2O, then H with H+ (acidic) or OH− (basic).
- Always verify charge balance at the end!
Method 2: Half-Reaction (Ion-Electron) Method
This method is very systematic. Follow these steps:
- Write the two half-reactions (oxidation and reduction separately).
- Balance all atoms EXCEPT O and H.
- Balance O by adding H2O.
- Balance H by adding H+ (acidic) or OH− (basic).
- Balance charge by adding electrons (e−).
- Multiply the half-reactions so that electrons cancel.
- Add the half-reactions and simplify.
Question: Balance using the half-reaction method in acidic medium: Cr2O72− + Fe2+ → Cr3+ + Fe3+
Solution:
Step 1: Write half-reactions:
Reduction: Cr2O72− → Cr3+
Oxidation: Fe2+ → Fe3+
Step 2: Balance atoms (except O and H):
Cr2O72− → 2Cr3+ (balance Cr)
Fe2+ → Fe3+ (already balanced)
Step 3: Balance O with H2O:
Cr2O72− → 2Cr3+ + 7H2O
Step 4: Balance H with H+:
Cr2O72− + 14H+ → 2Cr3+ + 7H2O
Step 5: Balance charge with electrons:
Left charge: −2 + 14 = +12; Right charge: 2(+3) = +6
Add 6e− to the left: Cr2O72− + 14H+ + 6e− → 2Cr3+ + 7H2O
For oxidation: Fe2+ → Fe3+ + e−
Step 6: Multiply oxidation half-reaction by 6:
6Fe2+ → 6Fe3+ + 6e−
Step 7: Add and cancel electrons:
Cr2O72− + 14H+ + 6e− + 6Fe2+ → 2Cr3+ + 7H2O + 6Fe3+ + 6e−
Balanced equation:
Cr2O72− + 6Fe2+ + 14H+ → 2Cr3+ + 6Fe3+ + 7H2O
Worked Example 3Question: Balance in basic medium: MnO4− + I− → MnO2 + I2
Solution:
Half-reactions:
Reduction: MnO4− → MnO2
Oxidation: I− → I2
Balance atoms: 2I− → I2
Balance O with H2O: MnO4− → MnO2 + 2H2O
Balance H with H+ (first balance as acidic): MnO4− + 4H+ → MnO2 + 2H2O
Balance charge: Left = −1 + 4 = +3; Right = 0. Add 3e− to left:
MnO4− + 4H+ + 3e− → MnO2 + 2H2O
Oxidation: 2I− → I2 + 2e−
Multiply to cancel electrons: LCM of 3 and 2 is 6.
2 × (MnO4− + 4H+ + 3e− → MnO2 + 2H2O)
3 × (2I− → I2 + 2e−)
Add: 2MnO4− + 8H+ + 6I− → 2MnO2 + 4H2O + 3I2
Now convert to basic medium: Add 8OH− to both sides to neutralize H+:
2MnO4− + 8H+ + 6I− + 8OH− → 2MnO2 + 4H2O + 3I2 + 8OH−
8H+ + 8OH− → 8H2O. Simplify: 8H2O on left, cancel 4 with right → 4H2O on left:
2MnO4− + 6I− + 4H2O → 2MnO2 + 3I2 + 8OH−
Practice Question 3: Balance in acidic medium: MnO4− + Sn2+ → Mn2+ + Sn4+
Answer:
Reduction: MnO4− + 8H+ + 5e− → Mn2+ + 4H2O
Oxidation: Sn2+ → Sn4+ + 2e−
LCM of 5 and 2 = 10. Multiply reduction by 2, oxidation by 5:
2MnO4− + 16H+ + 10e− → 2Mn2+ + 8H2O
5Sn2+ → 5Sn4+ + 10e−
Balanced: 2MnO4− + 5Sn2+ + 16H+ → 2Mn2+ + 5Sn4+ + 8H2O
Practice Question 4: Balance in basic medium: Cl2 → ClO− + Cl− (this is a disproportionation reaction where Cl2 is both oxidized and reduced!)
Answer:
This is a disproportionation — Cl goes from 0 to +1 (oxidation) and from 0 to −1 (reduction).
Reduction: Cl2 + 2e− → 2Cl−
Oxidation: Cl2 + 4OH− → 2ClO− + 2H2O + 2e−
(Balanced for basic: O with H2O, H with OH−, then charge with e−)
Add: 2Cl2 + 4OH− → 2Cl− + 2ClO− + 2H2O
Simplify: Cl2 + 2OH− → Cl− + ClO− + H2O
2.2 Electrolysis of Aqueous Solutions
2.2.1 Electrolytic Cells
Now let’s learn about using electrical energy to drive non-spontaneous chemical reactions. This process is called electrolysis, and the device used is called an electrolytic cell.
Key terms:
- Anode: The positive electrode where oxidation occurs.
- Cathode: The negative electrode where reduction occurs.
- Electrolyte: The substance (molten salt or solution) that conducts electricity through the movement of ions.
Can you see the difference from what we learned before? In electrolytic cells, the anode is positive and the cathode is negative. This is because an external power source (battery) forces electrons to flow in a particular direction.
Remember for electrolytic cells: Anode = Positive = Oxidation; Cathode = Negative = Reduction.
2.2.2 Preferential Discharge
Here is a very important question: when you have an aqueous solution, there are multiple ions present — the ions from the salt AND the ions from water (H+ and OH−). Which ion gets discharged (loses its charge) at each electrode?
The answer depends on the electrode potential (a measure of how easily an ion gains or loses electrons). There is a specific order called preferential discharge:
If halide ions (Cl−, Br−, I−) are present → halide is discharged preferentially over OH−.
If NO halide ions are present (only SO42−, NO3−, etc.) → OH− is discharged: 4OH− → O2 + 2H2O + 4e−
If the metal ion is below hydrogen in the reactivity series (Cu2+, Ag+, Au+, etc.) → metal ion is discharged.
If the metal ion is above hydrogen (Na+, K+, Ca2+, Mg2+, Zn2+, etc.) → H+ from water is discharged: 2H+ + 2e− → H2
| Position | Cations (cathode) | Anions (anode) |
|---|---|---|
| Discharged FIRST | Ag+, Cu2+, Au+ (below H2) | I−, Br−, Cl− (halides) |
| Discharged SECOND | H+ (from water) | OH− (from water) → O2 |
| NOT discharged | Na+, K+, Ca2+, Mg2+, Al3+ | SO42−, NO3−, CO32− |
- At cathode: metals below H2 in reactivity series are discharged; above H2, hydrogen gas is produced.
- At anode: halides (Cl−, Br−, I−) are discharged over OH−; otherwise O2 is produced from OH−.
- Sulfate (SO42−) and nitrate (NO3−) are NEVER discharged in aqueous solution.
- The concentration of ions and the nature of the electrode can sometimes affect which ion is discharged.
Practice Question 5: Predict the products at each electrode during the electrolysis of aqueous CuSO4 using inert electrodes.
Answer:
Ions present: Cu2+, SO42−, H+, OH−
Cathode (−): Cu2+ is below H2 in reactivity series → Cu2+ + 2e− → Cu(s) (copper deposits)
Anode (+): No halide present → OH− is discharged: 4OH− → O2(g) + 2H2O(l) + 4e−
Products: Copper at cathode, oxygen gas at anode.
Practice Question 6: Predict the products at each electrode during the electrolysis of aqueous NaCl using inert electrodes.
Answer:
Ions present: Na+, Cl−, H+, OH−
Cathode (−): Na+ is above H2 in reactivity series → H+ is discharged: 2H+ + 2e− → H2(g)
Anode (+): Cl− is a halide → Cl− is discharged: 2Cl− → Cl2(g) + 2e−
Products: Hydrogen gas at cathode, chlorine gas at anode. Na+ and OH− remain in solution (NaOH is produced).
2.2.3 Electrolysis of Some Selected Aqueous Solutions
Let’s now study specific examples in detail. This is heavily tested in exams!
(a) Electrolysis of Dilute H2SO4 (Acidified Water)
Ions present: H+, SO42−, OH−
Cathode: 2H+ + 2e− → H2(g)
Anode: 4OH− → O2(g) + 2H2O(l) + 4e−
Overall: 2H2O(l) → 2H2(g) + O2(g)
This is really the electrolysis of water! The ratio of H2 : O2 produced is 2 : 1 by volume.
(b) Electrolysis of Concentrated NaCl Solution (Brine)
Ions present: Na+, Cl−, H+, OH−
Cathode: 2H+ + 2e− → H2(g)
Anode: 2Cl− → Cl2(g) + 2e−
Na+ and OH− remain → NaOH solution is left behind.
Products: H2 (cathode), Cl2 (anode), NaOH (solution) — this is the chlor-alkali process!
(c) Electrolysis of CuSO4 Solution with Inert Electrodes
Ions: Cu2+, SO42−, H+, OH−
Cathode: Cu2+ + 2e− → Cu(s) (copper metal deposits)
Anode: 4OH− → O2(g) + 2H2O(l) + 4e−
As Cu2+ is removed and H+ builds up, the solution becomes increasingly acidic.
(d) Electrolysis of CuSO4 Solution with Copper Electrodes
This is a special and important case! The copper anode is not inert — it participates in the reaction!
Cathode: Cu2+ + 2e− → Cu(s) (copper deposits on cathode)
Anode: Cu(s) → Cu2+ + 2e− (copper anode dissolves!)
The concentration of CuSO4 stays constant! This process is called electroplating or electrorefining of copper. The impure copper anode loses copper, and pure copper deposits on the cathode.
Worked Example 4Question: During the electrolysis of aqueous CuSO4 using copper electrodes, a student noticed that the blue color of the solution did NOT change. Explain why.
Answer: With copper electrodes, Cu dissolves from the anode at the same rate that Cu2+ deposits at the cathode. The concentration of Cu2+ in solution remains constant, so the blue color (due to Cu2+) does not change.
- Dilute H2SO4 → electrolysis of water → H2 and O2 in 2:1 ratio.
- Concentrated NaCl → H2, Cl2, and NaOH (chlor-alkali process).
- CuSO4 with inert electrodes → Cu deposits (cathode), O2 evolves (anode), solution becomes acidic.
- CuSO4 with Cu electrodes → Cu transfers from anode to cathode; concentration stays constant.
- When the anode is made of the same metal as the cation in solution, the anode dissolves instead of OH− being discharged.
Practice Question 7: What happens to the pH of the solution during the electrolysis of aqueous CuSO4 using inert electrodes? Explain.
Answer: The pH decreases (solution becomes more acidic). At the anode, OH− is discharged (4OH− → O2 + 2H2O + 4e−), removing OH− from the solution. At the cathode, Cu2+ is discharged instead of H+, so H+ remains in the solution. The removal of OH− and retention of H+ makes the solution increasingly acidic.
2.3 Quantitative Aspects of Electrolysis
How much product will form during electrolysis? The answer was given by Michael Faraday through his two famous laws.
2.3.1 Faraday’s First Law of Electrolysis
Where:
m = mass of substance deposited (grams)
Z = electrochemical equivalent (g/C)
Q = quantity of electricity (coulombs, C)
I = current (amperes, A)
t = time (seconds, s)
We can also express this using Faraday’s constant (F). One faraday (F) = 96,500 C/mol, which is the charge carried by one mole of electrons.
Where M = molar mass (g/mol), n = number of electrons transferred per ion.
Worked Example 5Question: Calculate the mass of copper deposited when a current of 5.0 A is passed through CuSO4 solution for 30 minutes. (MCu = 63.5 g/mol)
Solution:
Step 1: Identify n: Cu2+ + 2e− → Cu, so n = 2
Step 2: Convert time: t = 30 min × 60 = 1800 s
Step 3: Calculate Q = I × t = 5.0 × 1800 = 9000 C
Step 4: Calculate mass:
Question: What current is needed to deposit 2.0 g of silver from AgNO3 solution in 20 minutes? (MAg = 108 g/mol)
Solution:
n = 1 (Ag+ + e− → Ag), t = 20 × 60 = 1200 s, m = 2.0 g
- Always convert time to seconds!
- Identify n correctly: Cu2+ → n = 2; Ag+ → n = 1; Al3+ → n = 3.
- 1 Faraday (F) = 96,500 C/mol = charge of 1 mole of electrons.
- Mass is directly proportional to current and time.
Practice Question 8: How long will it take to deposit 0.50 g of iron from FeCl2 solution using a current of 3.0 A? (MFe = 56 g/mol)
Answer:
Fe2+ + 2e− → Fe, so n = 2
m = (M × I × t) / (n × F)
0.50 = (56 × 3.0 × t) / (2 × 96500)
0.50 = 168t / 193000
t = (0.50 × 193000) / 168 = 96500 / 168 = 574 s = 9.57 minutes
2.3.2 Faraday’s Second Law of Electrolysis
Where E = equivalent weight = M/n (molar mass divided by number of electrons transferred).
Question: A current is passed through two cells in series: one containing AgNO3 and the other containing CuSO4. If 1.08 g of silver is deposited in the first cell, what mass of copper is deposited in the second cell? (MAg = 108, MCu = 63.5)
Solution:
Equivalent weight of Ag = 108/1 = 108 g/eq
Equivalent weight of Cu = 63.5/2 = 31.75 g/eq
Question: The same quantity of electricity deposited 0.32 g of copper from CuSO4 solution. What mass of zinc would be deposited from ZnSO4 solution by the same quantity? (MCu = 63.5, MZn = 65)
Solution:
ECu = 63.5/2 = 31.75; EZn = 65/2 = 32.5
- Applies when the SAME quantity of electricity passes through different cells connected in series.
- Equivalent weight E = M/n (NOT just molar mass!).
- For Ag+: E = 108/1 = 108; for Cu2+: E = 63.5/2 = 31.75; for Al3+: E = 27/3 = 9.
- The ratio of masses equals the ratio of equivalent weights.
Practice Question 9: When the same current was passed through solutions of AgNO3 and Al(NO3)3 in series for the same time, 2.16 g of silver was deposited. Calculate the mass of aluminum deposited. (MAg = 108, MAl = 27)
Answer:
EAg = 108/1 = 108; EAl = 27/3 = 9
mAl / mAg = EAl / EAg
mAl / 2.16 = 9 / 108 = 1/12
mAl = 2.16 / 12 = 0.18 g
2.4 Industrial Application of Electrolysis
Electrolysis is not just a laboratory technique — it has many important industrial applications. Let’s look at the key ones.
1. Extraction of Metals
Highly reactive metals (Na, K, Ca, Mg, Al) cannot be extracted by reduction with carbon. They are extracted by electrolysis of their molten salts.
Extraction of Aluminum (Hall-Héroult Process):
Aluminum is extracted by electrolysis of molten alumina (Al2O3) dissolved in molten cryolite (Na3AlF6) at about 950°C.
Cathode: Al3+ + 3e− → Al(l)
Anode: 2O2− → O2 + 4e−
The O2 reacts with the carbon anode: C + O2 → CO2. So the carbon anodes must be replaced periodically!
Extraction of Sodium (Down’s Cell):
Molten NaCl is electrolyzed at about 600°C.
Cathode: Na+ + e− → Na(l)
Anode: 2Cl− → Cl2(g) + 2e−
A steel gauze diaphragm prevents Na and Cl2 from recombining.
2. Electroplating
Electroplating is the process of coating a metal object with a thin layer of another metal by electrolysis. For example, coating iron with chromium to make it shiny and rust-resistant.
- The object to be plated is the cathode (negative electrode).
- The plating metal is the anode (positive electrode).
- The electrolyte contains ions of the plating metal.
Example — Chromium plating of iron:
- Cathode: Iron object
- Anode: Chromium metal
- Electrolyte: Cr2(SO4)3 solution
- Cathode reaction: Cr3+ + 3e− → Cr
- Anode reaction: Cr → Cr3+ + 3e−
3. Electrorefining of Metals
Impure metals can be purified by electrolysis. For example, copper is refined using CuSO4 solution.
- Anode: Impure copper
- Cathode: Pure copper (thin sheet)
- Electrolyte: CuSO4 solution with H2SO4
- As current flows, Cu dissolves from the impure anode and deposits on the pure cathode.
- Impurities either remain in solution or fall to the bottom as “anode mud” (which contains valuable metals like Ag and Au!).
4. Chlor-Alkali Industry
Electrolysis of concentrated NaCl solution (brine) produces three important chemicals:
- Hydrogen gas (cathode) — used in making ammonia, margarine
- Chlorine gas (anode) — used in water treatment, PVC production
- Sodium hydroxide (remains in solution) — used in soap, paper, aluminum production
- Al is extracted from molten Al2O3 in cryolite (Hall-Héroult process); carbon anodes are consumed.
- Na is extracted from molten NaCl (Down’s cell); uses a diaphragm.
- In electroplating: object = cathode, plating metal = anode.
- Electrorefining: impure metal = anode, pure metal = cathode.
- Chlor-alkali process: brine → H2 + Cl2 + NaOH.
Practice Question 10: Why is cryolite used in the extraction of aluminum?
Answer: Pure Al2O3 has a very high melting point (about 2050°C), which would require too much energy. Adding cryolite (Na3AlF6) lowers the melting point to about 950°C, making the process much more energy-efficient and cost-effective. Cryolite also improves the conductivity of the molten mixture.
Practice Question 11: In the electrorefining of copper, why do silver and gold collect as “anode mud” instead of dissolving?
Answer: Silver and gold are less reactive (more noble) than copper. They have higher reduction potentials, meaning they are NOT oxidized at the anode under the conditions used. While copper dissolves (Cu → Cu2+ + 2e−), Ag and Au simply fall off the anode and settle at the bottom as “anode mud.” This anode mud is actually a valuable by-product since it contains precious metals!
2.5 Voltaic Cells
So far we have studied electrolytic cells, where electrical energy drives a non-spontaneous reaction. Now let’s look at the opposite: voltaic (galvanic) cells, where a spontaneous chemical reaction produces electrical energy. This is how batteries work!
Key difference from electrolytic cell:
- In a voltaic cell, the anode is NEGATIVE and the cathode is POSITIVE.
- This is the OPPOSITE of an electrolytic cell!
Structure of a Voltaic Cell
A simple voltaic cell consists of:
- Two half-cells (each containing an electrode in an electrolyte solution)
- A salt bridge (or porous barrier) connecting the two half-cells
- An external wire connecting the two electrodes (where electrons flow)
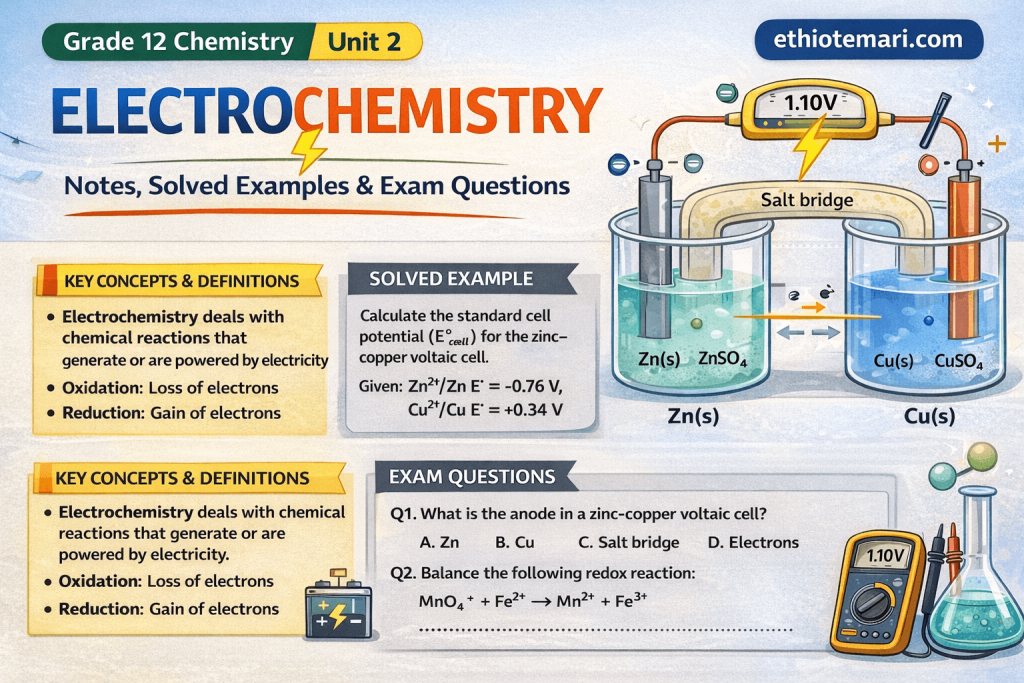
The Daniell Cell (Zn-Cu Cell)
This is the classic example of a voltaic cell:
Half-reactions:
Anode (oxidation): Zn(s) → Zn2+(aq) + 2e−
Cathode (reduction): Cu2+(aq) + 2e− → Cu(s)
Overall: Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s)
What does the salt bridge do?
- It completes the circuit by allowing ions to flow between the two half-cells.
- Without it, the solutions would build up charge and the reaction would stop.
- Anions (like NO3−) flow toward the anode (to balance the extra Zn2+ produced).
- Cations (like K+) flow toward the cathode (to replace the Cu2+ being consumed).
Electrode Potential and Cell Potential
Each half-cell has an electrode potential (E°), measured in volts. It tells us the tendency of a half-reaction to occur. Standard electrode potentials are measured against the Standard Hydrogen Electrode (SHE), which is assigned E° = 0.00 V.
| Half-Reaction | E° (V) |
|---|---|
| Li+ + e− → Li | −3.05 |
| K+ + e− → K | −2.93 |
| Na+ + e− → Na | −2.71 |
| Zn2+ + 2e− → Zn | −0.76 |
| Fe2+ + 2e− → Fe | −0.44 |
| 2H+ + 2e− → H2 | 0.00 (SHE) |
| Cu2+ + 2e− → Cu | +0.34 |
| Ag+ + e− → Ag | +0.80 |
| Au3+ + 3e− → Au | +1.50 |
The cell potential (E°cell) is calculated as:
Remember: Always subtract the anode potential from the cathode potential!
If E°cell > 0, the reaction is spontaneous (the cell will produce electricity). If E°cell < 0, the reaction is non-spontaneous.
Worked Example 9Question: Calculate the standard cell potential for the Daniell cell (Zn-Cu cell).
Solution:
Anode (oxidation): Zn → Zn2+ + 2e− → E° = −0.76 V
Cathode (reduction): Cu2+ + 2e− → Cu → E° = +0.34 V
Since E°cell = +1.10 V > 0, the reaction is spontaneous. This cell can produce electricity!
Worked Example 10Question: Will a voltaic cell made with Mg and Ag electrodes work? Calculate E°cell.
Solution:
Mg is more easily oxidized (more negative E°), so Mg is the anode and Ag is the cathode.
Anode: Mg → Mg2+ + 2e−; E° = −2.37 V
Cathode: Ag+ + e− → Ag; E° = +0.80 V
Yes! E°cell = 3.17 V > 0 — this cell definitely works and produces a large voltage.
- Voltaic cell: chemical energy → electrical energy (spontaneous reaction).
- Anode = negative (oxidation); Cathode = positive (reduction).
- Electrons flow from anode to cathode through the external wire.
- Salt bridge: completes the circuit; anions → anode, cations → cathode.
- E°cell = E°cathode − E°anode; must be positive for spontaneous reaction.
- The more negative E° value identifies the anode (more easily oxidized).
- Electrolytic cell: anode = positive, cathode = negative (OPPOSITE of voltaic!).
Practice Question 12: For a voltaic cell using Fe and Cu electrodes, calculate E°cell and write the overall cell reaction.
Answer:
Fe is more easily oxidized (E° = −0.44 V) than Cu (E° = +0.34 V).
Anode: Fe → Fe2+ + 2e− (E° = −0.44 V)
Cathode: Cu2+ + 2e− → Cu (E° = +0.34 V)
E°cell = 0.34 − (−0.44) = 0.78 V
Overall: Fe(s) + Cu2+(aq) → Fe2+(aq) + Cu(s)
Practice Question 13: A student sets up a cell with Zn|Zn2+ and Ag|Ag+ half-cells. Calculate E°cell. Which electrode is the anode?
Answer:
Zn has E° = −0.76 V and Ag has E° = +0.80 V.
Zn is more easily oxidized (more negative E°), so Zn is the anode and Ag is the cathode.
E°cell = 0.80 − (−0.76) = 1.56 V
Practice Question 14: Can a cell made with Ag|Ag+ and Au|Au3+ half-cells produce electricity? Explain.
Answer:
Ag: E° = +0.80 V; Au: E° = +1.50 V
Ag is more easily oxidized (less positive E°), so Ag is the anode and Au is the cathode.
E°cell = 1.50 − 0.80 = +0.70 V
Yes! Since E°cell = +0.70 V > 0, the cell is spontaneous and will produce electricity.
Overall: 3Ag(s) + Au3+(aq) → 3Ag+(aq) + Au(s)
Revision Notes — Exam Focus
Oxidation and Reduction at a Glance
| Oxidation | Reduction | |
|---|---|---|
| Electrons | Lost | Gained |
| Oxidation number | Increases | Decreases |
| Agent | Reducing agent | Oxidizing agent |
| Memory aid | LEO | GER |
Key Formulas
Important Definitions
- Oxidation: Loss of electrons or increase in oxidation number.
- Reduction: Gain of electrons or decrease in oxidation number.
- Reducing agent: The substance that is oxidized (causes another substance to be reduced).
- Oxidizing agent: The substance that is reduced (causes another substance to be oxidized).
- Electrolysis: Chemical change caused by passing electric current through an electrolyte.
- Electrolytic cell: Device that converts electrical energy to chemical energy (non-spontaneous).
- Voltaic cell: Device that converts chemical energy to electrical energy (spontaneous).
- Anode: Electrode where oxidation occurs.
- Cathode: Electrode where reduction occurs.
- Salt bridge: Connects two half-cells; allows ion flow to maintain electrical neutrality.
- Electroplating: Coating an object with a metal layer using electrolysis.
- Electrorefining: Purifying a metal by using it as the anode in electrolysis.
- Preferential discharge: Order in which ions are discharged at electrodes during electrolysis.
Electrolytic vs. Voltaic Cells — Quick Comparison
| Feature | Electrolytic Cell | Voltaic Cell |
|---|---|---|
| Energy conversion | Electrical → Chemical | Chemical → Electrical |
| Reaction type | Non-spontaneous | Spontaneous |
| Anode charge | Positive (+) | Negative (−) |
| Cathode charge | Negative (−) | Positive (+) |
| External source | Required (battery) | Not required |
| Salt bridge | Not always needed | Required |
Preferential Discharge — Quick Rules
Standard Electrode Potentials to Remember
| Half-Reaction | E° (V) |
|---|---|
| K+ + e− → K | −2.93 |
| Na+ + e− → Na | −2.71 |
| Mg2+ + 2e− → Mg | −2.37 |
| Al3+ + 3e− → Al | −1.66 |
| Zn2+ + 2e− → Zn | −0.76 |
| Fe2+ + 2e− → Fe | −0.44 |
| 2H+ + 2e− → H2 | 0.00 |
| Cu2+ + 2e− → Cu | +0.34 |
| Ag+ + e− → Ag | +0.80 |
| Au3+ + 3e− → Au | +1.50 |
Common Mistakes to Avoid
- Mistake 1: Confusing anode/cathode signs in electrolytic vs. voltaic cells. Electrolytic: anode = +, cathode = −. Voltaic: anode = −, cathode = +.
- Mistake 2: Forgetting to convert time to seconds in Faraday’s law calculations.
- Mistake 3: Using the wrong value of n (number of electrons). For Cu2+, n = 2; for Al3+, n = 3; for Ag+, n = 1.
- Mistake 4: In Faraday’s Second Law, using molar mass instead of equivalent weight (E = M/n).
- Mistake 5: Getting the wrong order in E°cell formula. It is ALWAYS E°cathode − E°anode, not the reverse!
- Mistake 6: Forgetting to balance charge when balancing half-reactions. Always check that total charge on left = total charge on right.
- Mistake 7: Thinking SO42− can be discharged at the anode. It CANNOT — only halides or OH− are discharged.
- Mistake 8: Forgetting to add OH− to BOTH sides when converting from acidic to basic medium in half-reaction balancing.
- Mistake 9: Forgetting that in CuSO4 with Cu electrodes, the anode dissolves (not O2 production).
- Mistake 10: Confusing reducing agent and oxidizing agent. The substance OXIDIZED is the REDUCING agent.
Challenge Exam Questions
These questions are designed to test your deep understanding. Try them all before checking the answers!
Section A: Multiple Choice Questions
Question 1: In the reaction 2Fe3+ + 2I− → 2Fe2+ + I2, which substance is the oxidizing agent?
(a) Fe3+ (b) I− (c) Fe2+ (d) I2
Answer: (a) Fe3+
Fe3+ is reduced to Fe2+ (gains electrons, oxidation number decreases from +3 to +2). The substance that is reduced is the oxidizing agent. I− is oxidized to I2 (it is the reducing agent).
Question 2: During the electrolysis of dilute Na2SO4 using inert electrodes, the products are:
(a) Na and O2 (b) H2 and O2 (c) Na and SO2 (d) H2 and SO2
Answer: (b) H2 and O2
Na+ is above H2 → H+ from water is discharged at cathode: 2H+ + 2e− → H2. SO42− is not a halide → OH− from water is discharged at anode: 4OH− → O2 + 2H2O + 4e−. This is essentially electrolysis of water.
Question 3: In a voltaic cell, electrons flow from:
(a) Cathode to anode through external circuit (b) Anode to cathode through external circuit
(c) Anode to cathode through the salt bridge (d) Salt bridge to the electrodes
Answer: (b) Anode to cathode through external circuit
In a voltaic cell, oxidation at the anode produces electrons. These electrons flow through the external wire from the anode (negative) to the cathode (positive) where reduction occurs. Ions flow through the salt bridge, NOT electrons.
Question 4: What is the oxidation number of Cr in Cr2O72−?
(a) +6 (b) +7 (c) +3 (d) +12
Answer: (a) +6
2x + 7(−2) = −2 → 2x − 14 = −2 → 2x = 12 → x = +6. Each Cr has oxidation number +6 in Cr2O72−.
Question 5: The function of a salt bridge in a voltaic cell is to:
(a) Provide a path for electron flow (b) Maintain electrical neutrality
(c) Increase the cell voltage (d) Prevent the reaction from occurring
Answer: (b) Maintain electrical neutrality
As the cell operates, Zn2+ builds up in the anode half-cell and Cu2+ is depleted in the cathode half-cell. Without a salt bridge, charge would build up and stop the reaction. The salt bridge allows ions to flow and maintain electrical neutrality. Electrons flow through the external wire, NOT the salt bridge.
Section B: Fill in the Blanks
Question 6: The mass of a substance deposited during electrolysis is __________ proportional to the quantity of electricity passed (Faraday’s First Law).
Answer: directly
Question 7: In the electrolysis of aqueous CuSO4 using copper electrodes, the __________ electrode decreases in mass and the __________ electrode increases in mass.
Answer: anode (decreases in mass as Cu dissolves); cathode (increases in mass as Cu deposits).
Question 8: One faraday equals __________ coulombs, which corresponds to __________ mole(s) of electrons.
Answer: 96,500 coulombs; 1 mole of electrons.
Question 9: In a voltaic cell, the __________ has the more negative standard electrode potential and serves as the site of __________.
Answer: anode; oxidation. The electrode with more negative E° is more easily oxidized, so it becomes the anode where oxidation occurs.
Question 10: During the electrolysis of concentrated NaCl solution, the three products are H2, Cl2, and __________.
Answer: NaOH (sodium hydroxide). H+ from water is reduced at the cathode (→ H2), Cl− is oxidized at the anode (→ Cl2), and Na+ and OH− remain in solution as NaOH.
Section C: Short Answer Questions
Question 11: Explain the difference between an electrolytic cell and a voltaic cell in terms of: (a) energy conversion, (b) spontaneity, (c) anode charge.
Answer:
(a) Energy conversion: Electrolytic cell converts electrical energy to chemical energy. Voltaic cell converts chemical energy to electrical energy.
(b) Spontaneity: Electrolytic cell drives a non-spontaneous reaction using external energy. Voltaic cell uses a spontaneous reaction to produce energy.
(c) Anode charge: In an electrolytic cell, the anode is positive (connected to + terminal of battery). In a voltaic cell, the anode is negative (source of electrons).
Question 12: Why is it important to use a diaphragm in the Down’s cell for sodium extraction?
Answer: In the Down’s cell, molten NaCl is electrolyzed to produce Na at the cathode and Cl2 at the anode. Sodium metal is highly reactive and would react violently with chlorine gas if they mixed: 2Na + Cl2 → 2NaCl. The steel gauze diaphragm separates the two products, preventing them from coming into contact and recombining.
Question 13: During the electrolysis of CuSO4 with inert electrodes, the solution gradually becomes acidic. Explain this observation using half-reactions.
Answer:
At the cathode: Cu2+ + 2e− → Cu (Cu2+ is removed from solution)
At the anode: 4OH− → O2 + 2H2O + 4e− (OH− is removed from solution)
Since OH− is being consumed but H+ is NOT being consumed (because Cu2+ is discharged instead of H+), the concentration of H+ increases relative to OH−. The solution becomes increasingly acidic (pH decreases).
Question 14: Define “disproportionation reaction” and give one example from this unit.
Answer: A disproportionation reaction is a redox reaction in which the same element is both oxidized AND reduced. Example: Cl2 + 2OH− → Cl− + ClO− + H2O. Here, Cl goes from 0 to −1 (reduction) and from 0 to +1 (oxidation) simultaneously.
Section D: Step-by-Step Calculation Questions
Question 15: Balance the following redox reaction in acidic medium using the half-reaction method: MnO4− + C2O42− → Mn2+ + CO2
Answer:
Reduction half: MnO4− → Mn2+
Balance Mn: done
Balance O: MnO4− → Mn2+ + 4H2O
Balance H: MnO4− + 8H+ → Mn2+ + 4H2O
Balance charge: −1 + 8 = +7 (left); +2 (right). Add 5e− to left:
MnO4− + 8H+ + 5e− → Mn2+ + 4H2O
Oxidation half: C2O42− → 2CO2
Balance C: done (2 on each side)
Balance O: done (4 on each side)
Balance charge: −2 (left); 0 (right). Add 2e− to right:
C2O42− → 2CO2 + 2e−
LCM of 5 and 2 = 10:
2 × (MnO4− + 8H+ + 5e− → Mn2+ + 4H2O)
5 × (C2O42− → 2CO2 + 2e−)
Balanced: 2MnO4− + 5C2O42− + 16H+ → 2Mn2+ + 10CO2 + 8H2O
Question 16: Calculate the mass of aluminum deposited when a current of 10.0 A is passed through molten Al2O3 for 2 hours. (MAl = 27 g/mol)
Answer:
Al3+ + 3e− → Al, so n = 3
t = 2 hours × 3600 = 7200 s
Q = I × t = 10.0 × 7200 = 72,000 C
m = (M × Q) / (n × F) = (27 × 72000) / (3 × 96500) = 1,944,000 / 289,500 = 6.71 g
Question 17: A current of 2.5 A was passed through a solution of AgNO3 for 45 minutes. Calculate: (a) the mass of silver deposited, (b) the volume of gas evolved at STP if the same current is passed through dilute H2SO4 for the same time. (MAg = 108; molar volume at STP = 22.4 L/mol)
Answer:
t = 45 × 60 = 2700 s; Q = 2.5 × 2700 = 6750 C
(a) Silver deposited: Ag+ + e− → Ag, n = 1
m = (108 × 6750) / (1 × 96500) = 729000 / 96500 = 7.56 g
(b) Volume of H2 from dilute H2SO4:
At cathode: 2H+ + 2e− → H2 (n = 2)
Moles of electrons = Q / F = 6750 / 96500 = 0.06995 mol
Moles of H2 = 0.06995 / 2 = 0.03498 mol
Volume of H2 = 0.03498 × 22.4 = 0.784 L
Question 18: The same quantity of electricity was passed through solutions of CuSO4 and AgNO3 in series. If 3.175 g of copper was deposited, calculate: (a) the mass of silver deposited, (b) the mass of zinc that would be deposited from ZnSO4 by the same quantity. (MCu = 63.5, MAg = 108, MZn = 65)
Answer:
ECu = 63.5/2 = 31.75; EAg = 108/1 = 108; EZn = 65/2 = 32.5
(a) Mass of silver:
mAg / mCu = EAg / ECu
mAg / 3.175 = 108 / 31.75
mAg = 3.175 × (108/31.75) = 3.175 × 3.402 = 10.80 g
(b) Mass of zinc:
mZn / mCu = EZn / ECu
mZn / 3.175 = 32.5 / 31.75
mZn = 3.175 × (32.5/31.75) = 3.175 × 1.0236 = 3.25 g
Question 19: A voltaic cell is constructed using the following half-cells: Ni|Ni2+ (E° = −0.25 V) and Pb|Pb2+ (E° = −0.13 V). (a) Identify the anode and cathode. (b) Calculate E°cell. (c) Write the overall cell reaction.
Answer:
(a) Ni has the more negative E° (−0.25 V), so Ni is more easily oxidized. Ni is the anode and Pb is the cathode.
(b) E°cell = E°cathode − E°anode = (−0.13) − (−0.25) = −0.13 + 0.25 = +0.12 V
(c) Overall reaction:
Anode: Ni → Ni2+ + 2e−
Cathode: Pb2+ + 2e− → Pb
Overall: Ni(s) + Pb2+(aq) → Ni2+(aq) + Pb(s)
Question 20: During the electroplating of a spoon with silver, a current of 0.5 A is passed through AgNO3 solution for 1 hour. (a) Calculate the mass of silver deposited. (b) If the spoon has a surface area of 50 cm2, what is the thickness of the silver coating? (Density of Ag = 10.5 g/cm3; MAg = 108)
Answer:
t = 1 × 3600 = 3600 s; Q = 0.5 × 3600 = 1800 C
(a) Mass of silver:
m = (108 × 1800) / (1 × 96500) = 194400 / 96500 = 2.015 g
(b) Thickness of coating:
Volume = mass / density = 2.015 / 10.5 = 0.1919 cm3
Thickness = Volume / surface area = 0.1919 / 50 = 0.00384 cm = 0.0384 mm
The silver coating is approximately 0.038 mm thick.
Question 21: Balance the following in basic medium: CrO42− + Fe2+ → Cr(OH)3 + Fe3+
Answer:
Reduction half: CrO42− → Cr(OH)3
Balance Cr: done
Balance O: CrO42− → Cr(OH)3 + H2O (check: 4 O on left; 3+1=4 O on right ✓)
Balance H: CrO42− + 5H+ → Cr(OH)3 + H2O
Balance charge: −2 + 5 = +3 (left); 0 (right). Add 3e− to left:
CrO42− + 5H+ + 3e− → Cr(OH)3 + H2O
Oxidation half: Fe2+ → Fe3+ + e−
LCM of 3 and 1 = 3:
1 × (CrO42− + 5H+ + 3e− → Cr(OH)3 + H2O)
3 × (Fe2+ → Fe3+ + e−)
Add: CrO42− + 5H+ + 3Fe2+ → Cr(OH)3 + H2O + 3Fe3+
Convert to basic: Add 5OH− to both sides:
CrO42− + 5H+ + 3Fe2+ + 5OH− → Cr(OH)3 + H2O + 3Fe3+ + 5OH−
5H+ + 5OH− → 5H2O. Combine and simplify:
CrO42− + 3Fe2+ + 5H2O → Cr(OH)3 + H2O + 3Fe3+ + 5OH−
Cancel 1 H2O:
CrO42− + 3Fe2+ + 4H2O → Cr(OH)3 + 3Fe3+ + 5OH−
Question 22: How many minutes are required to produce 5.6 L of H2 at STP by electrolysis of acidified water using a current of 5 A?
Answer:
Moles of H2 = 5.6 / 22.4 = 0.25 mol
2H+ + 2e− → H2, so 1 mol H2 requires 2 mol e− = 2F charge
Total charge needed = 0.25 × 2 × 96500 = 48,250 C
Q = I × t → 48,250 = 5 × t
t = 48,250 / 5 = 9,650 s = 9,650 / 60 = 160.8 minutes ≈ 161 minutes