Welcome dear student! In this unit, we will study several very important families of organic compounds that all contain oxygen. These compounds are everywhere in our daily lives — from the ethanol in beverages and the ether used as anesthesia, to the flavors in fruits (esters) and the fats in our food. Let’s learn them one by one.
6.1 Introduction
Oxygen is the most abundant element in the Earth’s crust, and it forms bonds with carbon in many ways, creating several important classes of organic compounds. In this unit, we will cover: alcohols, ethers, aldehydes, ketones, carboxylic acids, esters, fats and oils. Each family has a characteristic functional group that determines its chemical properties.
6.2 Alcohols and Ethers
6.2.1 Classification of Alcohols
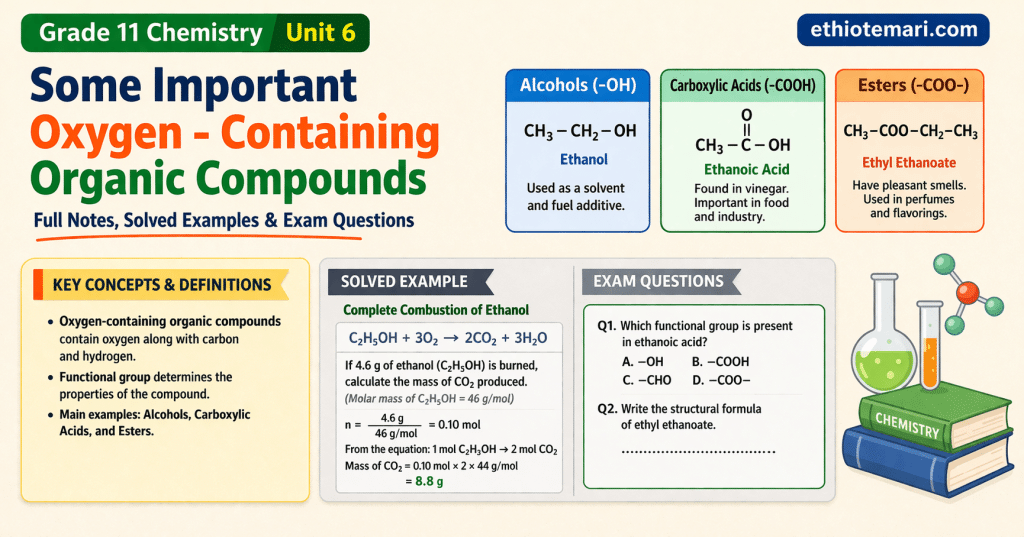
An alcohol is an organic compound containing the hydroxyl group (−OH) attached to an sp³ hybridized carbon atom. The general formula for alcohols is R−OH.
Alcohols are classified based on the number of alkyl groups attached to the carbon bearing the −OH group:
| Type | Structure | Description | Example |
|---|---|---|---|
| Primary (1°) | R−CH₂−OH | The −OH carbon is attached to only ONE other carbon | CH₃CH₂OH (ethanol) |
| Secondary (2°) | R₂CH−OH | The −OH carbon is attached to TWO other carbons | (CH₃)₂CHOH (2-propanol) |
| Tertiary (3°) | R₃C−OH | The −OH carbon is attached to THREE other carbons | (CH₃)₃COH (2-methyl-2-propanol) |
Alcohols are also classified by the number of −OH groups:
- Monohydric: One −OH group (e.g., ethanol)
- Dihydric (glycol): Two −OH groups (e.g., ethylene glycol)
- Trihydric (glycerol): Three −OH groups (e.g., glycerol)
6.2.2 Nomenclature of Alcohols
IUPAC rules for naming alcohols:
- Select the longest continuous carbon chain containing the −OH group.
- Number the chain from the end nearest the −OH group.
- Replace the −e ending of the alkane name with −ol.
- Indicate the position of the −OH group with a number.
- Name and number any substituents.
(a) CH₃CH₂CH₂OH (b) CH₃CH(OH)CH₃ (c) CH₃C(CH₃)₂OH
Solution:
(a) Three-carbon chain with −OH on C-1: 1-propanol
(b) Three-carbon chain with −OH on C-2: 2-propanol (common name: isopropyl alcohol)
(c) Three-carbon chain (propane) with −OH on C-2 and two methyl groups on C-2: 2-methyl-2-propanol (tertiary butyl alcohol)
6.2.3 Physical Properties of Alcohols
- Boiling points: Much higher than alkanes of similar molecular mass due to hydrogen bonding between −OH groups.
- Solubility: Lower alcohols (1−3 carbons) are soluble in water due to H-bonding with water. Solubility decreases as carbon chain length increases.
- State: Lower alcohols are liquids; higher members are waxy solids.
6.2.4 Preparation of Alcohols
Common methods of preparing alcohols:
- Hydration of alkenes: Addition of water to alkenes (with acid catalyst).
$$\text{CH}_2=\text{CH}_2 + \text{H}_2\text{O} \xrightarrow{\text{H}^+} \text{CH}_3\text{CH}_2\text{OH}$$ - Hydrolysis of alkyl halides: Reaction with aqueous NaOH.
$$\text{CH}_3\text{CH}_2\text{Br} + \text{NaOH}(aq) \rightarrow \text{CH}_3\text{CH}_2\text{OH} + \text{NaBr}$$ - Reduction of aldehydes and ketones: Using reducing agents like NaBH₄ or LiAlH₄.
$$\text{CH}_3\text{CHO} + 2[\text{H}] \rightarrow \text{CH}_3\text{CH}_2\text{OH}$$ - Fermentation: Sugars converted to ethanol by yeast enzymes.
$$\text{C}_6\text{H}_{12}\text{O}_6 \xrightarrow{\text{yeast}} 2\text{C}_2\text{H}_5\text{OH} + 2\text{CO}_2$$
6.2.5 Chemical Properties of Alcohols
1. Reaction with active metals: Alcohols react with Na, K, Mg to form alkoxides and hydrogen gas.
This reaction shows that alcohols are weakly acidic (much weaker than water).
2. Dehydration: Removal of water to form alkenes (with concentrated H₂SO₄, high temperature).
3. Oxidation:
- Primary alcohols → Aldehydes → Carboxylic acids (with strong oxidizing agents like K₂Cr₂O₇/H₂SO₄)
- Secondary alcohols → Ketones (cannot be further oxidized easily)
- Tertiary alcohols → No reaction with mild oxidizing agents (resist oxidation because no H on the −OH carbon)
(a) CH₃CH₂OH (b) (CH₃)₂CHOH (c) (CH₃)₃COH
Solution:
(a) Primary alcohol → aldehyde (ethanal, CH₃CHO) → with excess oxidant, carboxylic acid (ethanoic acid, CH₃COOH)
(b) Secondary alcohol → ketone (propanone, CH₃COCH₃)
(c) Tertiary alcohol → no reaction (no H atom on the carbon bearing −OH)
4. Esterification: Reaction with carboxylic acids to form esters (discussed in Section 6.5).
5. Reaction with hydrogen halides:
• Classification: 1°, 2°, 3° based on carbons attached to the −OH carbon.
• IUPAC: select chain with −OH, number from end nearest −OH, change −e to −ol.
• Higher boiling points than alkanes due to H-bonding.
• Oxidation: 1° → aldehyde → acid; 2° → ketone; 3° → no reaction.
• Dehydration with conc. H₂SO₄ at 170 °C gives alkene; at 140 °C gives ether.
• Na reacts with alcohols to produce H₂ gas — this distinguishes alcohols from alkanes.
1. Classify CH₃CH₂CH₂CH₂OH and (CH₃)₂C(OH)CH₂CH₃ as 1°, 2°, or 3° alcohols.
2. A compound gives a positive test with sodium metal (H₂ gas evolved) but does not react with acidified K₂Cr₂O₇. What type of alcohol could it be?
CH₃CH₂CH₂CH₂OH: The −OH is on C-1, which is attached to only one other carbon (C-2). → Primary (1°)
(CH₃)₂C(OH)CH₂CH₃: The −OH carbon is attached to three other carbons (two CH₃ and one CH₂). → Tertiary (3°)
Answer 2:
It could be a tertiary alcohol. Tertiary alcohols react with Na (to give H₂) because they have an −OH group, but they resist oxidation by K₂Cr₂O₇ because there is no hydrogen atom on the carbon bearing the −OH group.
6.2.6 Structure and Nomenclature of Ethers
An ether has the general structure R−O−R’, where R and R’ are alkyl or aryl groups. The −O− group is called the ether linkage.
Types:
- Simple (symmetrical) ethers: R = R’ (e.g., CH₃OCH₃, dimethyl ether)
- Mixed (unsymmetrical) ethers: R ≠ R’ (e.g., CH₃OC₂H₅, ethyl methyl ether)
Nomenclature: Name the two alkyl groups alphabetically, followed by “ether.” For IUPAC, the smaller group becomes “alkoxy” substituent on the larger chain.
Solution:
Common name: ethyl methyl ether (alphabetical order: ethyl before methyl)
IUPAC name: The larger chain is ethane (2 carbons). The CH₃O− group is methoxy. → methoxyethane
6.2.7 Physical Properties of Ethers
- Lower boiling points than alcohols of similar mass (no −OH, so no H-bonding).
- Slightly soluble in water (the oxygen can accept H-bonds from water).
- Good solvents for many organic reactions.
- Volatile and highly flammable.
6.2.8 Preparation of Ethers
Williamson ether synthesis: Reaction of an alkoxide ion with an alkyl halide.
Dehydration of alcohols: At 140 °C with H₂SO₄ (intermolecular dehydration).
6.2.9 Reactions of Ethers
Ethers are relatively unreactive (which is why they are good solvents). However:
- They can be cleaved by strong acids (e.g., concentrated HI).
- They are highly flammable (diethyl ether was used as an anesthetic but is dangerous due to flammability).
• General structure: R−O−R’. No −OH group, so no H-bonding between ether molecules.
• Lower b.p. than alcohols, but similar to alkanes of comparable mass.
• Williamson synthesis: alkoxide + alkyl halide → ether (best method).
• Dehydration at 140 °C gives ether; at 170 °C gives alkene.
• Ethers are relatively inert (unreactive) — this makes them good solvents.
1. Write the structure of diisopropyl ether.
2. Why does diethyl ether have a lower boiling point than 1-butanol even though they have similar molecular masses?
Diisopropyl ether = (CH₃)₂CH−O−CH(CH₃)₂
Both groups are isopropyl, so it is a symmetrical ether.
Answer 2:
1-Butanol has an −OH group that can form hydrogen bonds between molecules. These intermolecular H-bonds require significant energy to break, giving a higher boiling point. Diethyl ether has no −OH group — its oxygen cannot donate H-bonds to other ether molecules (it has no H attached to O). Ethers can only have weaker dipole-dipole interactions and London dispersion forces, resulting in a lower boiling point.
6.3 Aldehydes and Ketones
Both aldehydes and ketones contain the carbonyl group (C=O):
- Aldehyde: The carbonyl carbon is bonded to at least one H atom. General formula: RCHO (terminal C=O).
- Ketone: The carbonyl carbon is bonded to two carbon atoms. General formula: RCOR’ (internal C=O).
6.3.1 Nomenclature
Aldehydes: Replace −e with −al. The −CHO group is always at C-1, so no number is needed for the aldehyde group itself.
- Methanal (HCHO, formaldehyde)
- Ethanal (CH₃CHO, acetaldehyde)
- Propanal (CH₃CH₂CHO)
- Benzaldehyde (C₆H₅CHO)
Ketones: Replace −e with −one. The position of C=O must be numbered.
- Propanone (CH₃COCH₃, acetone) — simplest ketone
- Butanone (CH₃COCH₂CH₃)
- Benzophenone (C₆H₅COC₆H₅)
6.3.2 Physical Properties of Aldehydes and Ketones
- Cannot form H-bonds with each other (no −OH), so lower b.p. than alcohols of similar mass.
- Higher b.p. than ethers/alkanes due to stronger dipole-dipole forces from the polar C=O bond.
- Lower members (methanal, ethanal) are soluble in water — the C=O can accept H-bonds from water.
- Methanal is a gas; others are volatile liquids with distinctive odors.
Key chemical properties:
- Aldehydes are easily oxidized (reducing agents) — they reduce Fehling’s solution (red precipitate of Cu₂O) and Tollens’ reagent (silver mirror). Ketones do NOT give these tests.
- Both undergo nucleophilic addition reactions (e.g., with HCN, NaHSO₃).
- Both undergo condensation reactions (e.g., with hydrazine to form hydrazones).
• Both have C=O (carbonyl group). Aldehyde: terminal (RCHO). Ketone: internal (RCOR’).
• Aldehydes are reducing agents; ketones are NOT (this is the key distinction in tests).
• Fehling’s test: aldehyde → red precipitate; ketone → no change.
• Tollens’ test: aldehyde → silver mirror; ketone → no change.
• Neither can form H-bonds with themselves (no −OH), but can H-bond with water.
• Common examples: formaldehyde (preservative), acetone (nail polish remover, solvent).
1. Write the structure and IUPAC name of the ketone with 4 carbon atoms.
2. How would you distinguish between propanal and propanone using a chemical test?
CH₃COCH₂CH₃ — The C=O is on C-2 → butan-2-one (commonly called butanone or methyl ethyl ketone).
Answer 2:
Use Tollens’ reagent (ammoniacal silver nitrate). Add the reagent to each compound and warm.
• Propanal (an aldehyde): Will produce a silver mirror on the test tube (positive test).
• Propanone (a ketone): No change — no silver mirror forms (negative test).
Alternatively, Fehling’s solution can be used: propanal gives a red precipitate; propanone does not.
6.4 Carboxylic Acids
A carboxylic acid contains the carboxyl group (−COOH). General formula: RCOOH. The carboxyl group is the combination of a carbonyl (C=O) and a hydroxyl (−OH) on the same carbon.
6.4.1 Structure and Nomenclature
IUPAC naming: Select the longest chain containing the −COOH group. Replace −e with −oic acid. The carboxyl carbon is always C-1.
(a) HCOOH (b) CH₃COOH (c) CH₃CH₂COOH (d) CH₃CHClCOOH
Solution:
(a) Methanoic acid (common: formic acid — found in ant stings)
(b) Ethanoic acid (common: acetic acid — found in vinegar)
(c) Propanoic acid
(d) 2-chloropropanoic acid (Cl on C-2; COOH is C-1)
6.4.2 Physical Properties of Carboxylic Acids
- High boiling points — even higher than alcohols of similar mass due to strong H-bonding (two H-bonds per molecule: one as donor from −OH, one as acceptor through C=O).
- Lower members (1−4 carbons) are miscible with water due to H-bonding.
- Weak acids — they partially ionize in water: RCOOH ⇌ RCOO⁻ + H⁺
- Sharp, irritating odors (e.g., vinegar smell of ethanoic acid).
- Lower members are liquids; higher members are waxy solids.
6.4.3 Chemical Properties of Carboxylic Acids
1. Acidic nature: Carboxylic acids are weak acids. They react with metals, bases, and carbonates.
2. Esterification: Reaction with alcohols to form esters (discussed in Section 6.5).
3. Reduction: With strong reducing agents (LiAlH₄) to form primary alcohols.
4. Decarboxylation: Removal of CO₂ (heating sodium salt with soda lime, NaOH/CaO).
6.4.4 Preparation of Carboxylic Acids
- Oxidation of primary alcohols and aldehydes: Using K₂Cr₂O₇/H₂SO₄ or KMnO₄.
- Hydrolysis of esters: With acid or base (saponification).
- Carbonation of Grignard reagent: R MgBr + CO₂ → RCOOMgBr → RCOOH after acid workup.
6.4.5 Fatty Acids
Fatty acids are long-chain carboxylic acids (usually 12–22 carbons). They are classified as:
- Saturated: No C=C double bonds (e.g., stearic acid, C₁₇H₃₅COOH)
- Unsaturated: One or more C=C double bonds (e.g., oleic acid, C₁₇H₃₃COOH with one double bond; linoleic acid with two)
6.4.6 Uses of Carboxylic Acids
- Methanoic acid: rubber processing, textile dyeing, preservative.
- Ethanoic acid: vinegar, manufacture of cellulose acetate, esters, aspirin.
- Higher fatty acids: soap manufacture, food industry.
• Functional group: −COOH. IUPAC: −oic acid. COOH carbon is always C-1.
• Stronger acids than alcohols but still weak acids (pKa ≈ 4–5).
• Very high b.p. due to dimer formation through H-bonding.
• React with Na, NaOH, Na₂CO₃ — effervescence of H₂ or CO₂.
• Decarboxylation: sodium salt + soda lime → alkane (loses CO₂).
• Fatty acids: long-chain (C12–C22); saturated (no C=C) vs unsaturated (has C=C).
1. Write the equation for the reaction of ethanoic acid with sodium carbonate.
2. What is the product when sodium propanoate is heated with soda lime?
$$2\text{CH}_3\text{COOH} + \text{Na}_2\text{CO}_3 \rightarrow 2\text{CH}_3\text{COONa} + \text{H}_2\text{O} + \text{CO}_2\uparrow$$
Effervescence of CO₂ gas is observed — this is a test for carboxylic acids.
Answer 2:
Decarboxylation of sodium propanoate (CH₃CH₂COONa):
$$\text{CH}_3\text{CH}_2\text{COONa} + \text{NaOH} \xrightarrow{\text{CaO}, \Delta} \text{CH}_3\text{CH}_3 + \text{Na}_2\text{CO}_3$$
Product: ethane (C₂H₆). The carboxyl group is lost as CO₂ (which reacts with NaOH to form Na₂CO₃).
6.5 Esters
An ester has the general formula RCOOR’, formed by the reaction of a carboxylic acid with an alcohol.
6.5.1 Sources of Esters
Esters occur naturally in many fruits and flowers, where they are responsible for pleasant aromas. For example, isoamyl acetate gives banana flavor, and ethyl butanoate gives pineapple flavor.
6.5.2 Nomenclature
Name the alkyl group from the alcohol first, then the carboxylate part from the acid (replacing −oic acid with −oate).
Solution:
The alkyl part is ethyl (from ethanol, C₂H₅OH). The acid part is ethanoate (from ethanoic acid, CH₃COOH).
Name: ethyl ethanoate
6.5.3 Physical Properties
- Pleasant, fruity odors.
- Lower b.p. than the parent acids and alcohols (no H-bonding between ester molecules).
- Insoluble in water (no −OH for H-bonding with water).
- Good organic solvents.
6.5.4 Chemical Properties
Hydrolysis: Esters react with water (with acid or base catalyst) to reform the acid and alcohol.
6.5.5 Preparation of Esters
6.5.6 Uses of Esters
- Flavoring agents in food and beverages.
- Perfumes and cosmetics (fragrant odors).
- Solvents for paints, varnishes, and lacquers.
- Plasticizers and polyester fibers.
• General formula: RCOOR’. Formed from carboxylic acid + alcohol.
• Naming: alkyl (from alcohol) + carboxylate (from acid, −oate ending).
• Fischer esterification: acid + alcohol + conc. H₂SO₄ → ester + water (reversible!).
• Saponification: ester + NaOH → carboxylate salt + alcohol (base hydrolysis).
• Pleasant fruity odors; used in flavorings and perfumes.
• Hydrolysis is the reverse of esterification.
1. Write the equation for the formation of propyl methanoate from methanoic acid and propan-1-ol.
2. What is saponification? Write a general equation.
$$\text{HCOOH} + \text{CH}_3\text{CH}_2\text{CH}_2\text{OH} \xrightarrow{\text{conc. H}_2\text{SO}_4, \Delta} \text{HCOOCH}_2\text{CH}_2\text{CH}_3 + \text{H}_2\text{O}$$
Methanoic acid + propan-1-ol → propyl methanoate + water
Answer 2:
Saponification is the base-catalyzed hydrolysis of an ester to produce the sodium salt of a carboxylic acid (soap) and an alcohol.
$$\text{RCOOR}’ + \text{NaOH} \rightarrow \text{RCOONa} + \text{R}’\text{OH}$$
The carboxylate salt (RCOONa) is the soap. This reaction is the basis of soap manufacture.
6.6 Fats and Oils
6.6.1 Sources of Fats and Oils
Fats are obtained from animal sources (e.g., butter, tallow, lard). Oils are obtained from plant sources (e.g., olive oil, palm oil, sunflower oil). Chemically, both are triglycerides (triacylglycerols).
6.6.2 Structure of Fats and Oils
A triglyceride is an ester formed from glycerol (propane-1,2,3-triol, 3 −OH groups) and three fatty acid molecules.
• Fats: Contain mostly saturated fatty acids → solid at room temperature. Animal sources.
• Oils: Contain mostly unsaturated fatty acids (C=C bonds) → liquid at room temperature. Plant sources.
• The C=C bonds in oils create kinks in the fatty acid chains, preventing close packing → lower melting point → liquid.
6.6.3 Physical Properties of Fats and Oils
- Fats: solid at room temperature, higher melting points.
- Oils: liquid at room temperature, lower melting points.
- Both are insoluble in water (nonpolar triglyceride molecules).
- Less dense than water (float on water).
6.6.4 Hardening of Oils
Oils can be converted to fats by hydrogenation — adding hydrogen across the C=C double bonds using a nickel catalyst:
This process converts liquid oils to semi-solid fats (e.g., making margarine from vegetable oil). Partial hydrogenation is common in the food industry.
6.6.5 Rancidity
Rancidity is the unpleasant smell and taste that develops in fats and oils when they are exposed to air (oxidation) or moisture (hydrolysis). There are two types:
- Oxidative rancidity: Oxidation of unsaturated bonds by atmospheric oxygen, producing aldehydes and ketones with foul odors.
- Hydrolytic rancidity: Hydrolysis of triglycerides by moisture/enzymes, releasing free fatty acids (e.g., butanoic acid smells rancid).
Prevention of rancidity:
- Storing in airtight containers (exclude oxygen and moisture).
- Adding antioxidants (e.g., vitamin E, BHT, BHA).
- Refrigeration (slows oxidation).
- Using inert gas (N₂) packaging.
• Both are triglycerides (esters of glycerol + 3 fatty acids).
• Fats = saturated fatty acids, solid, animal source.
• Oils = unsaturated fatty acids (C=C), liquid, plant source.
• Hardening = hydrogenation (add H₂ across C=C, Ni catalyst).
• Rancidity: oxidative (air, O₂) or hydrolytic (moisture). Prevent by antioxidants, airtight storage, refrigeration.
• Saponification of fats/oils produces soap.
1. Why are oils liquid at room temperature while fats are solid?
2. What is the purpose of adding antioxidants to food products containing fats?
Oils contain unsaturated fatty acids with C=C double bonds. These double bonds create “kinks” or bends in the fatty acid chains, preventing the molecules from packing closely together. Weak intermolecular forces result in lower melting points → liquid at room temperature. Fats contain saturated fatty acids with straight chains that pack tightly, allowing stronger London dispersion forces → higher melting points → solid.
Answer 2:
Antioxidants prevent oxidative rancidity. They react with oxygen and free radicals before the oxygen can oxidize the unsaturated fatty acid chains in the oils/fats. Without oxidation, the formation of foul-smelling aldehydes and ketones is prevented, extending the shelf life of the food product.
Quick Revision Notes — Exam Focus
1. Functional Groups Summary
| Compound Class | Functional Group | General Formula | Example |
|---|---|---|---|
| Alcohol | −OH (hydroxyl) | R−OH | CH₃CH₂OH |
| Ether | −O− (ether linkage) | R−O−R’ | CH₃OCH₃ |
| Aldehyde | −CHO (carbonyl, terminal) | RCHO | CH₃CHO |
| Ketone | −CO− (carbonyl, internal) | RCOR’ | CH₃COCH₃ |
| Carboxylic acid | −COOH (carboxyl) | RCOOH | CH₃COOH |
| Ester | −COO− (ester linkage) | RCOOR’ | CH₃COOCH₃ |
2. Boiling Point Order (similar molecular mass)
3. Classification of Alcohols
4. Key Reactions Summary
5. Identification Tests
| Test | Alcohol | Aldehyde | Ketone | Carboxylic Acid | Ether |
|---|---|---|---|---|---|
| Na metal | H₂ gas ✓ | ✗ | ✗ | H₂ gas ✓ | ✗ |
| Tollens’ | ✗ | Ag mirror ✓ | ✗ | ✗ | ✗ |
| Fehling’s | ✗ | Red ppt ✓ | ✗ | ✗ | ✗ |
| Na₂CO₃ | ✗ | ✗ | ✗ | CO₂ ✓ | ✗ |
6. Common Mistakes to Avoid
❌ Forgetting that −CHO carbon is always C-1 (no number needed for aldehydes).
❌ Confusing oxidation products: 1° alcohol → aldehyde (NOT ketone).
❌ Saying ketones give Fehling’s/Tollens’ test — they do NOT.
❌ Forgetting that dehydration at 140 °C gives ether, 170 °C gives alkene.
❌ Writing the wrong ester name — alkyl from alcohol comes FIRST.
❌ Confusing fats (saturated, solid) with oils (unsaturated, liquid).
❌ Forgetting that saponification requires NaOH (base), not just water.
❌ Saying ethers have H-bonding — they do not (no −OH).
Challenge Exam Questions
Test yourself thoroughly! Try each question before checking the answer.
Section A: Multiple Choice Questions
A) CH₃CH₂CH₂OH B) (CH₃)₂CHOH C) (CH₃)₃COH D) CH₃CH₂CH(OH)CH₃
(CH₃)₃COH: The −OH carbon is attached to three other carbons (three CH₃ groups), making it tertiary. A has 1° (−OH carbon attached to 1 carbon), B has 2° (attached to 2 carbons), D has 2° (attached to 2 carbons).
A) Diethyl ether B) Ethylene C) Ethanal D) Ethanoic acid
At 170 °C with conc. H₂SO₄, ethanol undergoes intramolecular dehydration to form ethylene (CH₂=CH₂). At 140 °C, it would form diethyl ether (intermolecular dehydration). The temperature determines the product.
A) Propanone B) Propanoic acid C) Propanal D) Propan-1-ol
Propanal is an aldehyde. Aldehydes are reducing agents and reduce Tollens’ reagent (ammoniacal Ag⁺) to metallic silver, forming a silver mirror. Propanone (ketone) does not. Propanoic acid and propan-1-ol do not give this test.
A) Butyric acid B) Butanoic acid C) Propanoic acid D) Butanal
The chain has 4 carbons with −COOH at C-1. IUPAC: butanoic acid (from butane, replacing −e with −oic acid). Butyric acid is the common name, not IUPAC.
A) It breaks the ester bonds B) It saturates C=C double bonds
C) It removes glycerol D) It adds water molecules
Hydrogenation adds H₂ across the C=C double bonds in unsaturated fatty acids of oils, converting them to saturated fatty acids. Saturated chains pack more tightly, raising the melting point and converting liquid oil to semi-solid fat.
Section B: Fill in the Blanks
Section C: Short Answer Questions
Carboxylic acids form dimers — two acid molecules linked by TWO hydrogen bonds (the −OH of one acts as donor to the C=O of the other, and vice versa). This effectively doubles the molecular mass for intermolecular purposes. Alcohols form single H-bonds between molecules. The dimer structure of carboxylic acids, held together by two strong H-bonds, requires more energy to break, resulting in higher boiling points than alcohols of comparable molecular mass.
Use Tollens’ reagent (ammoniacal silver nitrate).
• Ethanol (alcohol): No reaction — no silver mirror forms.
• Ethanal (aldehyde): Silver mirror forms on the test tube (Ag⁺ reduced to Ag⁰ by the aldehyde).
Alternatively, use Fehling’s solution:
• Ethanol: No change.
• Ethanal: Red precipitate of Cu₂O forms.
A third option: Add acidified K₂Cr₂O₇. Ethanol (1° alcohol) will turn the orange solution green (Cr⁶⁺ reduced to Cr³⁺). Ethanal will also be oxidized further, but the test is less clear-cut than Tollens’/Fehling’s.
Ethers are preferred because they are relatively chemically inert (unreactive). Unlike alcohols, ethers do not have an acidic −OH group, so they don’t react with active metals, bases, or many other reagents that might interfere with the desired reaction. Additionally, ethers can dissolve both polar and nonpolar substances, making them versatile solvents. However, their extreme flammability is a safety concern.
Section D: Structured/Calculation Questions
(a) CH₃CH₂OH + Na →
(b) CH₃CHO + Tollens’ reagent →
(c) CH₃COOH + CH₃OH (conc. H₂SO₄) →
(d) CH₃COOCH₂CH₃ + NaOH(aq) →
(e) CH₃CH₂COONa + NaOH/CaO (heat) →
(a) $$2\text{CH}_3\text{CH}_2\text{OH} + 2\text{Na} \rightarrow 2\text{CH}_3\text{CH}_2\text{ONa} + \text{H}_2\uparrow$$
(b) $$\text{CH}_3\text{CHO} + 2[\text{Ag}(\text{NH}_3)_2]^+ + 3\text{OH}^- \rightarrow \text{CH}_3\text{COO}^- + 2\text{Ag}\downarrow + 4\text{NH}_3 + 2\text{H}_2\text{O}$$
(Silver mirror formed)
(c) $$\text{CH}_3\text{COOH} + \text{CH}_3\text{OH} \xrightarrow{\text{conc. H}_2\text{SO}_4, \Delta} \text{CH}_3\text{COOCH}_3 + \text{H}_2\text{O}$$
(Methyl ethanoate)
(d) $$\text{CH}_3\text{COOCH}_2\text{CH}_3 + \text{NaOH} \rightarrow \text{CH}_3\text{COONa} + \text{CH}_3\text{CH}_2\text{OH}$$
(Saponification: sodium ethanoate + ethanol)
(e) $$\text{CH}_3\text{CH}_2\text{COONa} + \text{NaOH} \xrightarrow{\text{CaO}, \Delta} \text{CH}_3\text{CH}_3 + \text{Na}_2\text{CO}_3$$
(Decarboxylation: ethane formed)
(a) Identify compounds A, B, and C.
(b) What type of alcohol is A? Explain your reasoning.
(c) Write equations for all reactions.
(a) A = 2-methyl-2-propanol ((CH₃)₃COH)
B = 2-methylpropene ((CH₃)₂C=CH₂)
C = 2-bromo-2-methylpropane ((CH₃)₃CBr)
(b) A is a tertiary alcohol. Evidence: it gives H₂ with Na (confirms −OH group, so it is an alcohol, not an ether), but does not react with K₂Cr₂O₇ (tertiary alcohols resist oxidation because there is no H atom on the carbon bearing the −OH group).
(c) $$(\text{CH}_3)_3\text{COH} \xrightarrow{170°\text{C}, \text{H}_2\text{SO}_4} (\text{CH}_3)_2\text{C}=\text{CH}_2 + \text{H}_2\text{O}$$
$$(\text{CH}_3)_2\text{C}=\text{CH}_2 + \text{HBr} \rightarrow (\text{CH}_3)_3\text{CBr}$$
(a) Identify X, Y, and Z.
(b) Explain the difference in boiling points between X and Y.
(c) Write the structure of Z and explain the test results.
(a) X = ethanal (CH₃CHO) — aldehyde (positive Tollens’)
Y = propanone (CH₃COCH₃) — ketone (negative Tollens’)
Z = 2-methyl-2-propanol ((CH₃)₃COH) — tertiary alcohol
(b) Although propanone (Y, M=58) has a higher molecular mass than ethanal (X, M=44), the main reason for the higher b.p. of Y is that propanone has two electron-donating CH₃ groups that increase the polarity of the C=O bond, leading to stronger dipole-dipole interactions compared to ethanal (which has only one CH₃ group). Also, the molecular mass difference contributes.
(c) Z = (CH₃)₃COH. Positive test with Na confirms the −OH group (alcohol, not ether). Negative test with K₂Cr₂O₇ confirms it is tertiary (no H on −OH carbon to be oxidized). Note: Z could also be 2-butanol (2° alcohol) based on the formula, but a 2° alcohol WOULD be oxidized by K₂Cr₂O₇. Since no reaction occurs, it must be 3°.
(a) Ethanol to ethanoic acid
(b) Ethanol to ethene
(c) Ethanoic acid to ethyl ethanoate
(d) Ethyl ethanoate to ethanol
(e) Ethanoic acid to ethane
(a) $$\text{CH}_3\text{CH}_2\text{OH} \xrightarrow{\text{K}_2\text{Cr}_2\text{O}_7/\text{H}_2\text{SO}_4} \text{CH}_3\text{CHO} \xrightarrow{\text{excess K}_2\text{Cr}_2\text{O}_7} \text{CH}_3\text{COOH}$$
(Oxidation: alcohol → aldehyde → acid)
(b) $$\text{CH}_3\text{CH}_2\text{OH} \xrightarrow{\text{conc. H}_2\text{SO}_4, 170°\text{C}} \text{CH}_2=\text{CH}_2 + \text{H}_2\text{O}$$
(Dehydration)
(c) $$\text{CH}_3\text{COOH} + \text{CH}_3\text{CH}_2\text{OH} \xrightarrow{\text{conc. H}_2\text{SO}_4, \Delta} \text{CH}_3\text{COOCH}_2\text{CH}_3 + \text{H}_2\text{O}$$
(Esterification)
(d) $$\text{CH}_3\text{COOCH}_2\text{CH}_3 + \text{NaOH}(aq) \rightarrow \text{CH}_3\text{COONa} + \text{CH}_3\text{CH}_2\text{OH}$$
(Saponification/base hydrolysis)
(e) $$\text{CH}_3\text{COONa} + \text{NaOH} \xrightarrow{\text{CaO}, \Delta} \text{CH}_4 + \text{Na}_2\text{CO}_3$$
(Decarboxylation of sodium ethanoate gives methane)
(a) Diethyl ether (C₄H₁₀O) has a much lower boiling point than butan-1-ol (C₄H₁₀O).
(b) Methanoic acid is a stronger acid than ethanoic acid.
(c) Fats are solid while oils are liquid at room temperature.
(a) Butan-1-ol has an −OH group that forms intermolecular hydrogen bonds between molecules. These strong H-bonds require significant energy to overcome, giving a high boiling point. Diethyl ether has no −OH group — its oxygen cannot donate H-bonds to other ether molecules. Ether molecules interact only through weaker dipole-dipole forces and London dispersion forces, resulting in a much lower boiling point despite the same molecular formula.
(b) Methanoic acid (HCOOH) is stronger because the H atom directly attached to the carboxyl group has a slight electron-withdrawing inductive effect on the carboxylate anion, stabilizing it. In ethanoic acid (CH₃COOH), the CH₃ group has a weak electron-donating effect that destabilizes the carboxylate anion (CH₃COO⁻) slightly, making deprotonation less favorable. Also, in methanoic acid, there is no electron-donating alkyl group to destabilize the conjugate base.
(c) Fats contain saturated fatty acids with straight carbon chains that pack closely together, allowing strong London dispersion forces between chains → high melting point → solid. Oils contain unsaturated fatty acids with C=C double bonds that create kinks in the chains, preventing close packing → weaker intermolecular forces → lower melting point → liquid.
Step 1 — Test with Na₂CO₃ solution:
Add a small amount of each compound to Na₂CO₃ solution.
• Compound C produces effervescence (CO₂ bubbles) → C is ethanoic acid (carboxylic acids react with carbonates).
• A and B show no effervescence → both are not carboxylic acids.
Step 2 — Tollens’ test (or Fehling’s test) on A and B:
Add Tollens’ reagent to A and B separately and warm.
• Compound A gives a silver mirror → A is ethanal (aldehyde — reducing agent).
• Compound B shows no silver mirror → B is propanone (ketone — not a reducing agent).
Result: A = ethanal, B = propanone, C = ethanoic acid.
The triglyceride structure:
Glycerol connects via ester bonds to three fatty acids:
• Position 1: stearoyl (saturated, C₁₇H₃₅CO−)
• Position 2: oleoyl (unsaturated, C₁₇H₃₃CO−, C=C at C-9,10)
• Position 3: oleoyl (unsaturated, C₁₇H₃₃CO−, C=C at C-9,10)
General structure: Stearoyl-O-CH₂ | Oleoyl-O-CH | Oleoyl-O-CH₂ (where the middle carbon of glycerol bears the second oleoyl group).
This is an oil because two of the three fatty acids are unsaturated (oleic acid has C=C bonds). The C=C bonds create kinks in the fatty acid chains, preventing close packing and resulting in a lower melting point — liquid at room temperature.